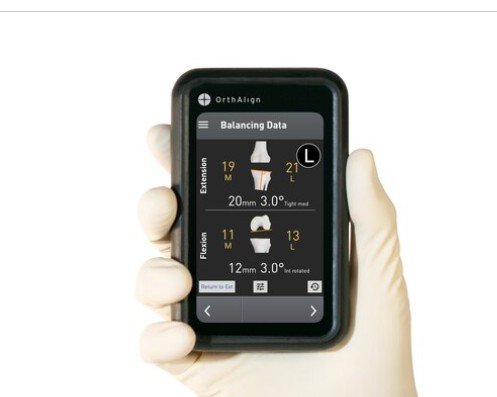
OrthAlign Secures CE Mark for Navigation Solutions: A New Era for Handheld Orthopedic Technology
OrthAlign, Inc., a pioneer in handheld orthopedic enabling technology, has made a significant stride in expanding its footprint in the European market. The company announced on Monday that it has successfully received the CE Mark under the European Union Medical Device Regulation (EU MDR) for its innovative portfolio of navigation solutions. This certification not only underscores the reliability and safety of OrthAlign’s technologies but also paves the way for their commercialization across a variety of European markets.
OrthAlign’s acclaimed products, including the Lantern and OrthAlign Plus platforms, are now CE marked and officially available for sale in Europe. The certification signifies that the company has met European standards for medical devices, reinforcing its commitment to providing surgeons with advanced orthopedic solutions. This achievement is vital for the company as it seeks to gain a larger foothold in a region where demand for cutting-edge orthopedic technologies continues to rise.
Streamlined Technology for Surgeons
One of the primary advantages of OrthAlign’s products is their design ethos, focused on accessibility and ease of implementation. According to Eric Timko, the CEO of OrthAlign, the technology was created to facilitate seamless integration into the surgical workflow. With no requirement for pre-operative imaging, minimal training, and rapid product availability, OrthAlign’s solutions allow surgeons to adopt new technologies without affecting their existing workflows. This essential feature is particularly appealing to busy surgical teams looking to enhance patient outcomes while maintaining efficiency.
The absence of pre-operative imaging sets OrthAlign apart from many competing technologies, which often require extensive planning and imaging to function effectively. By eliminating these barriers, OrthAlign enables surgeons to focus more on the surgical procedure and patient care rather than on complicated technological requirements. The swift adoption of these navigation solutions can lead to more timely surgeries and, ultimately, improve the overall quality of care provided to patients.
Addressing the Surgical Needs of Today
As the healthcare landscape continues to evolve, there is an increasing demand for adaptable and efficient medical technologies. OrthAlign’s handheld navigation solutions cater precisely to this need by offering tools that are not only effective but also user-friendly. Surgeons often find themselves pressed for time, and the ability to integrate new tools into their practice without lengthy training sessions is invaluable. OrthAlign’s commitment to simplicity ensures that their technology can be adopted quickly and effectively.
Furthermore, the innovative nature of the Lantern and OrthAlign Plus platforms means they can provide real-time data and insights during surgical procedures, enhancing decision-making capabilities for orthopedic surgeons. This data-driven approach not only helps in improving surgical precision but also contributes to better patient outcomes, which is a critical metric in today’s medical environment.
The Broader Impact on Orthopedic Surgery
The receipt of the CE Mark represents more than just a regulatory milestone; it signifies a broader shift towards the adoption of technological advancements in orthopedic surgery. As healthcare systems in Europe continue to prioritize efficiency and patient care, tools like those offered by OrthAlign will likely play a pivotal role in transforming surgical practices. The integration of innovative technologies into the operating room can lead to improved accuracy in orthopedic procedures, reducing recovery times and enhancing patient satisfaction.
OrthAlign’s solutions are not merely technological enhancements; they represent a paradigm shift in how orthopedic surgeries are conducted. With a focus on user-friendly designs and operational efficiency, these navigating solutions have the potential to become standard practice in operating rooms across Europe and beyond.
Looking Ahead: Future Opportunities for OrthAlign
With the successful launch of CE-marked products in Europe, OrthAlign is poised to explore further opportunities in the global market. This initial success opens the door for additional innovations and advancements in orthopedic enabling technology. The company is likely to continue developing its product lines, with the aim of positioning itself as a leader in the orthopedic navigation segment worldwide.
Expanding its influence in European markets also may encourage OrthAlign to invest more in partnerships and collaborations with healthcare institutions. By forging strategic alliances, the company can harness collective expertise and expand its reach further, potentially leading to enhanced product offerings and improved patient outcomes.
Conclusion: A Milestone for OrthAlign, Inc.
In summary, OrthAlign, Inc.’s receipt of the CE Mark for its handheld orthopedic navigation solutions marks a pivotal moment in the company’s journey and underscores its commitment to advancing surgical practices. With innovative products like the Lantern and OrthAlign Plus now available in European markets, surgeons can embrace new technologies that improve workflow efficiencies and patient care.
As the company builds on this success, it is set to drive further advancements in orthopedic surgery, making it easier for surgeons to incorporate cutting-edge technology into their practice. The potential benefits of this technology are expansive, signaling a promising future for both OrthAlign and the orthopedic community as a whole.
This article is based on reporting from www.ocbj.com.
The original version of the story can be found on their website.
Original Source:
www.ocbj.com
Image Credit: www.ocbj.com ·
View image